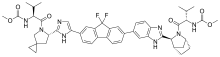
LEDIPASVIR, GS 5885
CAS 1256388-51-8
Methyl N-[(2S)-1-[(6S)-6-[5-[9,9-Difluoro-7-[2-[(1S,2S,4R)-3-[(2S)-2-(methoxycarbonylamino)-3-methylbutanoyl]-3-azabicyclo[2.2.1]heptan-2-yl]-3H-benzimidazol-5-yl]fluoren-2-yl]-1H-imidazol-2-yl]-5-azaspiro[2.4]heptan-5-yl]-3-methyl-1-oxobutan-2-yl]carbamate
Ledipasvir (formerly GS-5885) is an experimental drug for the treatment of hepatitis C being developed by Gilead Sciences.[1] It is currently in Phase III clinical trials.[2] It is being studied in combination with other direct-acting antiviral agents that interfere with HCV replication.
Ledipasvir is an inhibitor of the hepatitis C virus HCV NS5A protein.
Ledipasvir is being tested in interferon-free regimens with other direct-acting antiviral agents for hepatitis C.
Data presented at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013 showed that a triple regimen of the nucleotide analogue inhibitor sofosbuvir, ledipasvir, and ribavirin produced a 12-week post-treatment sustained virological response (SVR12) rate of 100% for both treatment-naive patients and prior non-responders with HCV genotype 1.[3][4] Gilead is developing a sofosbuvir + ledipasvir coformulation that is being tested with and without ribavirin.
- ^ "Ledipasvir". United States Adopted Name.
- ^ "GS-5885". Gilead Sciences.
- ^ ELECTRON: 100% Suppression of Viral Load through 4 Weeks’ Post-treatment for Sofosbuvir + Ledipasvir (GS-5885) + Ribavirin for 12 Weeks in Treatment-naïve and -experienced Hepatitis C Virus GT 1 Patients. Gane, Edward et al. 20th Conference on Retroviruses and Opportunistic Infections. March 3–6, 2013. Abstract 41LB.
- ^ CROI 2013: Sofosbuvir + Ledipasvir + Ribavirin Combo for HCV Produces 100% Sustained Response. Highleyman, Liz. HIVandHepatitis.com. 4 March 2013.
Hepatitis C is recognized as a chronic viral disease of the liver which is characterized by liver disease. Although drugs targeting the liver are in wide use and have shown
effectiveness, toxicity and other side effects have limited their usefulness. Inhibitors of hepatitis C virus (HCV) are useful to limit the establishment and progression of infection by HCV as well as in diagnostic assays for HCV.
The compound (l-{3-[6-(9,9-dif uoro-7-{2-[5-(2-methoxycarbonylamino-3-methyl- butyryl)-5-aza-spiro[2.4]hept-6-yl]-3H-imidazol-4-yl}-9H-fluoren-2-yl)-lH-benzoimidazol-2- yl]-2-aza-bicyclo[2.2.1]heptane-2-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester, also known as ledipasvir, designated herein as Compound I, is known to be an effective anti-HCV agent, as described for example in WO 2010/132601. A synthesis of compound I is disclosed in U.S. Patent No. 8,088,368


acetone solvate . 1H NMR (400 MHz, DMSO-^, δ): 12.29 (s, 0.1H), 12.19 (d, J=4.0 Hz, 1H), 12.14 (s, 0.2H), 11.85 (s, 1H), 8.10 (s, 0.1H), 8.08 (s, 1H), 8.01 (s, 0.1H), 7.963 (m, 1H), 7.955 (s, 1H), 7.89 (d, J=6.4 Hz, 1H), 7.87 (s, 1H), 7.83 (dd, J=8.4, 2.4 Hz, 1H), 7.79 (dd, J=7.2, 2.8 Hz, 1H), 7.78-7.90 (misc., 0.9H), 7.70 (s, 1H), 7.61 (d, J=8.4 Hz, 1H), 7.55 (s, 1H), 7.51 (dd, J=8.8, 1.6 Hz, 1H), 7.44 (m, 0.1H), 7.31 (d, J=8.4 Hz, 1H), 7.21 (d, J=8.4 Hz, 1H), 6.91 (d, J=8.0 Hz, 0.2H), 6.77 (m, 0.2H), 5.34 (d, J=7.6 Hz, 0.1H), 5.20 (dd, J=8.0, 5.2 Hz, 1H), 5.18 (m, 0.1H), 4.88 (s, 0.1H), 4.67 (d, J=6.4 Hz, 1H), 4.55 (s, 1H), 4.17 (dd, J=8.0, 8.0 Hz, 1H), 4.10 (m, 0.2H), 4.01 (dd, J=8.4, 8.0 Hz, 1H), 3.97 (m, 0.1H), 3.82 (d, J=9.6 Hz, 1H), 3.77 (s, 0.2H), 3.71 (d, J=9.6 Hz, 1H), 3.554 (s, 3H), 3.548 (s, 3H), 3.43 (s, 0.4H), 3.20 (d, J=7.6 Hz, 0.3H), 2.77 (s, 0.1H), 2.66 (s, 1H), 2.41 (d, J=8.8 Hz, 1H), 2.22 (dd, J=12.4, 8.0 Hz, 1H), 2.13 (m, 0.4H), 2.08 (s, 6H), 2.05 (dd, J=13.2, 5.2 Hz, 1H), 1.99 (m, 2H), 1.92 (m, 1H), 1.77 (m, 2H), 1.61 (m, 0.3H), 1.56 (m, 1H), 1.46 (d, J=9.2 Hz, 1H), 1.33 (d, J=10.0 Hz, 0.1H), 0.97 (dd, J=6.4, 2.0 Hz, 3H), 0.93 (d, J=6.8 Hz, 3H), 0.88 (d, J=6.4 Hz, 3H), 0.87 (d, J=6.4 Hz, 3H), 0.80-1.05 (misc., 2H), 0.70 (m, 1H), 0.59 (m, 2H), 0.54 (m, 1H), 0.33 (m, 0.1H). HRMS-ESI+: [M + H]+ calcd for C49H5506N8F2, 889.4207; found, 889.4205.
....................................................................................................





..............................................................
https://www.google.co.in/patents/US8088368?dq=US+8088368&hl=en&sa=X&ei=0aq5UsyWAcjyrQfZmYHADw&ved=0CDcQ6AEwAA

\\
4-Methylene-pyrrolidine-1,2-dicarboxylic acid 1-tert-butyl ester (10.0 g, 44 mmol) was dissolved in MeOH (75 mL) at room temperature and HCl (4M in dioxane, 75 mL) was added. Stirring at room temperature was continued for 4 hours. All volatiles were removed in vacuo and a beige solid was obtained.
The crude material was suspended in DCM (100 mL) and N-Methyl morpholine (13.3 g, 132 mmol) was added. The mixture was cooled to 0° C. and benzyl chloroformate (8.26 g, 48.4 mmol) was added while stirring. After 30 minutes, the reaction was warmed to room temperature and the solution was washed with water and aqueous HCl (1M). The solution was dried over sodium sulfate. Filtration and evaporation of solvents gave crude product, which was purified by silica gel chromatography (eluent: EtOAc/hexanes) to yield the product (10.2 g). LCMS-ESI+: calc'd for C15H17NO4: 275.3 (M+). Found: 276.4 (M+H+).
5-aza-spiro[2.4]heptanes-5,6-dicarboxylic acid benzyl ester: An oven-dried 3-neck round bottom flask was equipped with a nitrogen inlet adaptor and a 250 mL addition funnel. The third neck was sealed with a septum. The flask was charged with a stir bar, dichlorormethane (120 mL) and diethyl zinc (1.0 M in hexane, 118 mL, 118 mmol) then cooled to 0° C. in an ice bath. The addition funnel was charged with dichloromethane (40 mL) and trifluoroacetic acid (9.1 mL, 118 mmol). After the diethyl zinc solution had cooled to 0° C. (about 25 minutes), the trifluoroacetic acid solution was added dropwise over 20 minutes to the stirred reaction mixture. After stirring for another 20 minutes at 0° C., diiodomethane (9.5 mL, 118 mmol) was added slowly over 4 minutes. After another 20 minutes, 4-methylene-pyrrolidine-1,2-dicarboxylic acid 1-benzyl ester 2-methyl ester (8.10 g, 29.4 mmol) was added in 30 mL dichloromethane by cannula. The flask containing 4-methylene-pyrrolidine-1,2-dicarboxylic acid 1-benzyl ester 2-methyl ester was then rinsed with another 10 mL dichloromethane and this solution was also transferred to the reaction mixture by cannula. The reaction mixture was allowed to warm to RT and stirred for 110 hours (about 5 days) after which the reagents were quenched with saturated aqueous ammonium chloride (˜150 mL). The contents of the flask were slowly poured into a 2 L sep funnel containing saturated aqueous sodium bicarbonate (˜800 mL). The aqueous phase was extracted three times with 300 mL ethyl acetate. The combined organics were dried over magnesium sulfate and concentrated to provide the crude material. The crude material was dissolved in 3:1:1 THF/water/acetone (165 mL) then treated with N-methylmorpholine-N-oxide (3.45 g, 29.4 mmol) and osmium tetroxide (4 wt % in water, 5 mL, 0.818 mmol). After stirring at RT for 7 h, the reagents were quenched with 1 M aqueous sodium thiosulfate (˜100 mL). The contents of the flask were then poured into a 1 L sep funnel containing water (˜300 mL). The aqueous phase was extracted three times with 300 mL dichloromethane. The combined organics were dried over magnesium sulfate and concentrated. The crude residue was purified by silica column chromatography (5% to 45% EtOAc/hexane) to provide 5-aza-spiro[2.4]heptane-5,6-dicarboxylic acid 5-benzyl ester 6-methyl ester as a clear oil (5.54 g, 19.15 mmol, 65%) as a clear oil. 1H NMR (CDCl3) δ 7.36-7.29 (m, 5H), 5.21-5.04 (m, 2H), 4.56-4.47 (m, 1H), 3.75 (s, 1.5H), 3.60 (m, 1.5H), 03.51-3.37 (m, 2H), 2.32-2.25 (m, 1H), 1.87-1.80 (m, 1H), 0.64-0.51 (m, 4H).
5-Aza-spiro[2.4]heptane-5,6-dicarboxylic acid 5-benzyl ester 6-methyl ester (244 mg, 0.840 mmol) was dissolved in THF (2.0 mL)/MeOH (1.5 mL) An aqueous solution of LiOH (35.5 mg, 0.84 mmol) was added and stirring at room temperature was continued. After 3 hours, the reaction was neutralized with aqueous HCl (1M) and the organic solvents were removed in vacuo. The crude mixture was diluted with water and EtOAc and the organic layer was collected. All volatiles were removed in vacuo and the crude acid was used without further purification. LCMS-ESI+: calc'd for C15H17NO4: 275.3 (M+). Found: 276.3 (M+H+).
2,7-Dibromo-fluoren-9-one (4.0 g, 11.8 mmol) was suspended in deoxofluor (12 mL) at room temperature and EtOH (4 drops) was added. The stirred suspension was heated at T=90° C. for 24 hours (CAUTION: Use of deoxofluor at elevated temperatures, as described above, is strongly discouraged as rapid and violent exotherms may occur). The reaction was cooled to room temperature and poured onto ice containing sodium bicarbonate. A solid formed and was collected via filtration. The crude material was taken into EtOAc and was washed with aqueous HCl (1M) and brine. The solution was dried over sodium sulfate. Filtration and evaporation of solvents gave crude product, which was purified by silica gel chromatography (eluent: EtOAc/hexanes) to yield the product 2,7-Dibromo-9,9-difluoro-9H-fluorene (3.2 g). 19F-NMR: 282 MHz, (dmso-d6) δ: −111.6 ppm.
Before using the material in the next step, it was exposed as a solution in EtOAc to charcoal.
2,7-Dibromo-9,9-difluoro-9H-fluorene (372 mg, 1.04 mmol), Pd(PPh3)4 (30.0 mg, 0.026 mmol), PdCl2(PPh3)2 (18.2 mg, 0.026 mmol), As(PPh3)3 (5.0 mg) were dissolved in dioxane (10 mL) under an argon atmosphere. Ethoxyvinyl-tributyl tin (376.4 mg, 1.04 mmol) was added. The mixture was heated for 140 minutes at 85° C. (oil bath). The reaction was cooled to room temperature. N-bromo succinimide (177 mg, 1.0 mmol) was added followed by water (2 mL). The reaction was stirred at room temperature for 3 hours, after which the majority of the dioxane was removed in vacuo. The crude reaction mixture was diluted with EtOAc and was washed with water. All volatiles were removed in vacuo. Toluene was added and all volatiles were removed in vacuo for a second time. The crude material was dissolved in DMF/MeCN (2 mL, 1:1) at room temperature. A solution of N-Cbz-4-cyclopropyl (L) Proline (0.84 mmol) and DIEA (268 mg, 2.08 mmol) in MeCN (2 mL) was added and stirring at room temperature was continued. After 14 hours, most of the MeCN was removed in vacuo and the crude reaction mixture was diluted with EtOAc. The mixture was washed with aqueous HCl (1M), aqueous LiCl solution (5%), brine, and was dried over sodium sulfate. Filtration and evaporation of solvents gave the crude reaction product, which was purified via silica gel chromatography (eluent: EtOAc/hexanes) to yield the product 5-Aza-spiro[2.4]heptane-5,6-dicarboxylic acid 5-benzyl ester 6-[2-(7-bromo-9,9-difluoro-9H-fluoren-2-yl)-2-oxo-ethyl]ester (176 mg). LCMS-ESI+: calc'd for C30H24BrF2NO5: 596.4 (M+). Found: 595.2/597.2 (M+H+).
5-Aza-spiro[2.4]heptane-5,6-dicarboxylic acid 5-benzyl ester 6-[2-(7-bromo-9,9-difluoro-9H-fluoren-2-yl)-2-oxo-ethyl]ester (172 mg, 0.293 mmol) was dissolved in m-xylenes (6.0 mL). Ammonium acetate (226 mg, 2.93 mmol) was added and the reaction was stirred at 140° C. for 60 minutes under microwave conditions. The reaction was cooled to room temperature and all volatiles were removed in vacuo. The crude material was purified via silica gel chromatography (eluent: EtOAc/hexanes) to yield the product 6-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-aza-spiro[2.4]heptane-5-carboxylic acid benzyl ester (80.3 mg). LCMS-ESL': calc'd for C30H24BrF2N3O2: 576.4 (M+). Found: 575.2/577.2 (M+H+).
6-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-aza-spiro[2.4]heptane-5-carboxylic acid benzyl ester (800 mg, 1.38 mmol) was dissolved in DCM (15 mL) and HBr in AcOH (37%, 2 mL) was added and stirring at room temperature was continued. After 180 minutes, the suspension was diluted with hexanes and the solid was collected via filtration and was washed with hexanes and subjected to vacuum. The crude material was used in the next step without further purification. The crude material was dissolved in DMF (4.0 mL) and DIEA (356 mg, 2.76 mmol) was added. A solution of 2-(L)-Methoxycarbonylamino-3-methyl-butyric acid (242 mg, 1.38 mmol), HATU (524 mg, 1.38 mmol) and DIEA (178 mg, 1.38 mmol) in DMF (1 mL) was added. The reaction was stirred at room temperature. After 50 minutes, the reaction was diluted with EtOAc and was washed with aqueous bicarbonate solution, aqueous LiCl solution (5%), brine, and was dried over sodium sulfate. Filtration and removal of solvents in vacuo gave the crude material, which was purified by silica gel chromatography (eluent: EtOAc/hexanes) to yield the slightly impure product (1-{6-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-aza-spiro[2.4]heptane-5-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester (878 mg). LCMS-ESI+: calc'd for C29H29BrF2N4O3: 599.5 (M+); Found: 598.5/600.5 (M+H+).
(1-{6-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-5-aza-spiro[2.4]heptane-5-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester (840 mg, 1.4 mmol), 3-[6-(4,4,5,5-Tetramethyl-[1,3,2]dioxaborolan-2-yl)-1H-benzoimidazol-2-yl]-2-aza-bicyclo[2.2.1]heptane-2-carboxylic acid tert-butyl ester (615 mg, 1.4 mmol), Pd(PPh3)4 (161 mg, 0.14 mmol), K2CO3 (579 mg, 4.2 mmol), were dissolved in DME (15 mL)/water (3 mL) under an argon atmosphere. The mixture was heated for 120 minutes at 85-90° C. (oil bath). After 120 minutes additional boronate ester (61 mg, 0.14 mmol) was added and heating was continued. After 3 hours, the reaction was cooled to room temperature. Most of the DME was removed in vacuo and the crude reaction mixture was diluted with EtOAc. The mixture was washed with brine and was dried over sodium sulfate. Filtration and evaporation of solvents gave the crude reaction product, which was purified via silica gel chromatography (eluent: EtOAc/hexanes) to yield the product 3-[6-(9,9-Difluoro-7-{2-[5-(2-methoxycarbonylamino-3-methyl-butyryl)-5-aza-spiro[2.4]hept-6-yl]-3H-imidazol-4-yl}-9H-fluoren-2-yl)-1H-benzoimidazol-2-yl]-2-aza-bicyclo[2.2.1]heptane-2-carboxylic acid tert-butyl ester (878 mg). LCMS-ESI+: calc'd for C47H51F2N7O5: 831.9 (M+). Found: 832.7 (M+H+).
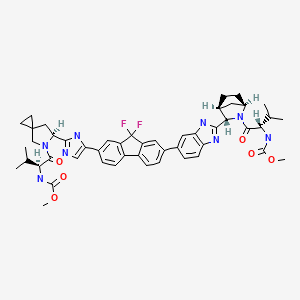
3-[6-(9,9-Difluoro-7-{2-[5-(2-methoxycarbonylamino-3-methyl-butyryl)-5-aza-spiro[2.4]hept-6-yl]-3H-imidazol-4-yl}-9H-fluoren-2-yl)-1H-benzoimidazol-2-yl]-2-aza-bicyclo[2.2.1]heptane-2-carboxylic acid tert-butyl ester (115 mg, 0.138 mmol) was dissolved in DCM (2 mL) and HCl in dioxane (4M, 2 mL) was added and stirring at room temperature was continued. After 20 minutes, all volatiles were removed in vacuo. The crude material was used in the next step without further purification. The crude material was dissolved in DMF (1.5 mL) and DIEA (53.4 mg, 0.414 mmol) was added. A solution of 2-(L) Methoxycarbonylamino-3-methyl-butyric acid (24.2 mg, 0.138 mmol), HATU (52.4 mg, 0.138 mmol) and DIEA (17.8 mg, 0.138 mmol) in DMF (1 mL) was added. The reaction was stirred at room temperature. After 20 minutes, the reaction was diluted with EtOAc and was washed with aqueous bicarbonate solution, aqueous LiCl solution (5%), brine, and was dried over sodium sulfate. Filtration and removal of solvents in vacuo gave the crude material, which was purified by RP-HPLC (eluent: water/MeCN w/0.1% TFA) to yield the product (1-{3-[6-(9,9-Difluoro-7-{2-[5-(2-methoxycarbonylamino-3-methyl-butyryl)-5-aza-spiro[2.4]hept-6-yl]-3H-imidazol-4-yl}-9H-fluoren-2-yl)-1H-benzoimidazol-2-yl]-2-aza-bicyclo[2.2.1]heptane-2-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester (76 mg). LCMS-ESI+: calc'd for C49H54F2N8O6: 888.9 (M+). Found: 890.0 (M+H+).
1H-NMR: 300 MHz, (dmso-d6) δ: 8.20-7.99 (m, 8H), 7.73 (s, 2H), 7.37-7.27 (m, 2H), 5.25 (dd, J=7.2 Hz, 1H), 4.78 (s, 1H) 4.54 (s, 1H), 4.16 (m, 1H), 4.02 (m, 1H), 3.87 (m, 1H), 3.74 (m, 1H), 3.55 (s, 3H), 3.53 (s, 3H), 2.75 (m, 1H), 2.25 (m, 2H), 2.09-2.04 (m, 2H), 1.88-1.79 (m, 2H), 1.54 (m, 1H), 0.94-0.77 (m, 15H) 0.63 (m, 4H) ppm. 19F-NMR: 282 MHz, (dmso-d6) δ: −109.1 ppm [−74.8 ppm TFA]
https://www.google.co.in/patents/US8088368?dq=US+8088368&hl=en&sa=X&ei=0aq5UsyWAcjyrQfZmYHADw&ved=0CDcQ6AEwAA

2-(5-{9,9-Difluoro-7-[2-(2-Boc-2-aza-bicyclo[2.2.1]hept-3-yl)-3H-benzoimidazol-5-yl]-9H-fluoren-2-yl}-1H-imidazol-2-yl)-pyrrolidine-1-carboxylic acid tert-butyl ester: A mixture of 2-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1H-imidazol-2-yl]-pyrrolidine-1-carboxylic acid tert-butyl ester (324 mg, 0.627 mmol), 3-[6-(4,4,5,5-Tetramethyl-[1,3,2]dioxaborolan-2-yl)-1H-benzoimidazol-2-yl]-2-aza-bicyclo[2.2.1]heptane-2-carboxylic acid tert-butyl ester (1.1 eq., 304 mg), [1,1′ bis(diphenylphosphino)ferrocene]dichloropalladium(II)(3%, 15 mg), tetrakis(triphenylphosphine)palladium (3%, 22 mg) and potassium carbonate (3.3 eq., 285 mg) in 10 mL DME and 3 mL water was heated to 90° C. under Argon for 3 hours. The reaction mixture was cooled and diluted with ethyl acetate and washed with saturated sodium bicarbonate solution. The organic layer was dried (MgSO4), concentrated and purified by flash column chromatography (silica gel, 20 to 100% ethyl acetate/hexane) to give 2-(5-{9,9-Difluoro-7-[2-(2-Boc-2-aza-bicyclo[2.2.1]hept-3-yl)-3H-benzoimidazol-5-yl]-9H-fluoren-2-yl}-1H-imidazol-2-yl)-pyrrolidine-1-carboxylic acid tert-butyl ester (361 mg, yield 77%). LCMS-ESI−: calc'd for C43H46F2N6O4: 748.86. Found: 749.2 (M+H+).
(1-{2-[5-(9,9-Difluoro-7-{2-[2-(2-methoxycarbonylamino-3-methyl-butyryl)-2-aza-bicyclo[2.2.1]hept-3-yl]-3H-benzoimidazol-5-yl}-9H-fluoren-2-yl)-1H-imidazol-2-yl]-pyrrolidine-1-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester (Example DK): 4N HCl in dioxane (2 mL) was added to 2-(5-{9,9-Difluoro-7-[2-(2-Boc-2-aza-bicyclo[2.2.1]hept-3-yl)-3H-benzoimidazol-5-yl]-9H-fluoren-2-yl}-1H-imidazol-2-yl)-pyrrolidine-1-carboxylic acid tert-butyl ester (361 mg, 0.482 mmol) and the reaction mixture was stirred at room temperature for 4 hours. The reaction mixture was concentrated and dried overnight under vacuum. The residue was dissolved in DMF (5 mL) and to this solution was added 2-Methoxycarbonylamino-3-methyl-butyric acid (2 eq., 169 mg), diisopropyl ethylamine (6 eq., 0.5 mL), followed by HATU (2 eq., 367 mg). Reaction mixture was stirred at 0° C. for 30 minutes. The reaction mixture was dissolved in ethyl acetate and washed with saturated sodium bicarbonate solution. The organic layer was dried (MgSO4), concentrated and purified by flash column chromatography (silica gel, 0 to 20% MeOH/ethyl acetate), followed by preparative reverse phase HPLC (GEMINI, 5 to 100% ACN/H2O+0.1% TFA). Product was lyophilized to give (1-{2-[5-(9,9-Difluoro-7-{2-[2-(2-methoxycarbonylamino-3-methyl-butyryl)-2-aza-bicyclo [2.2.1]hept-3-yl]-3H-benzoimidazol-5-yl}-9H-fluoren-2-yl)-1H-imidazol-2-yl]-pyrrolidine-1-carbonyl}-2-methyl-propyl)-carbamic acid methyl ester (285 mg, 59%).
1H-NMR: 300 MHz, (CD3OD-d4) δ: 8.05-7.82 (m, 9H), 5.40-5.22 (m, 2H), 4.72 (m, 1H), 4.39 (d, 1H), 4.239d, 1H), 4.17 (m, 1H), 3.91 (m, 2H), 3.62 (d, 6H), 2.98 (m, 1H), 2.58 (m, 1H), 2.37-2.18 (m, 4H), 2.18-1.92 (m, 4H), 1.80 (m, 2H), 1.09-0.85 (m, 12H). 19F-NMR: 300 MHz, (CD3OD-d4) δ: −112.88. LCMS-ESI+: calc'd for C47H52F2N8O6 862.96. Found: 863.5 (M+H+).
................................................................................................................

